High-altitude hypoxia exposure inhibits erythrophagocytosis by inducing macrophage ferroptosis in the spleen
Figures
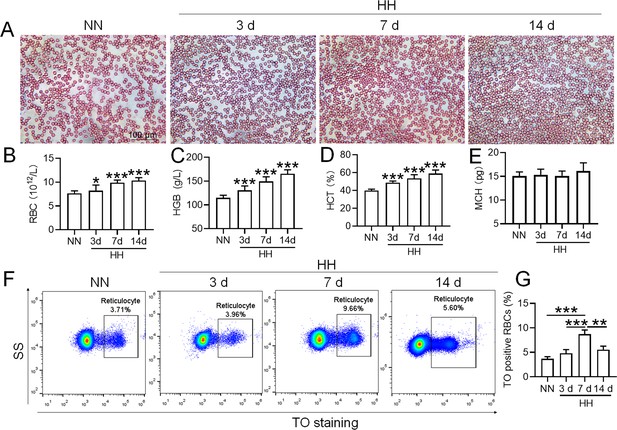
HH exposure promotes the induction of erythrocytosis in mice.
C57BL/6 mice were subjected to either normobaric normoxia (NN) or hypobaric hypoxia (HH) conditions for durations of 3, 7, and 14 days. After these treatments, blood samples were collected for a comprehensive analysis. (A) Morphological evaluation of RBCs was conducted via blood smear examination (Wright staining). Routine hematological assessments were performed, encompassing RBCs counts (B), hemoglobin (HGB) levels (C), hematocrit (HCT) percentages (D), and mean corpuscular hemoglobin (MCH) content (E). (F) Flow cytometric analysis was employed to identify TO-positive cells, indicative of reticulocytes, in the whole blood samples. (G) The proportions of TO-positive RBCs in the blood were depicted in bar graphs. The data are presented as means ± SEM for each group (n=5 per group). Statistical significance is denoted by * p<0.05, ** p<0.01, *** p<0.001, relative to the NN group or as specified.
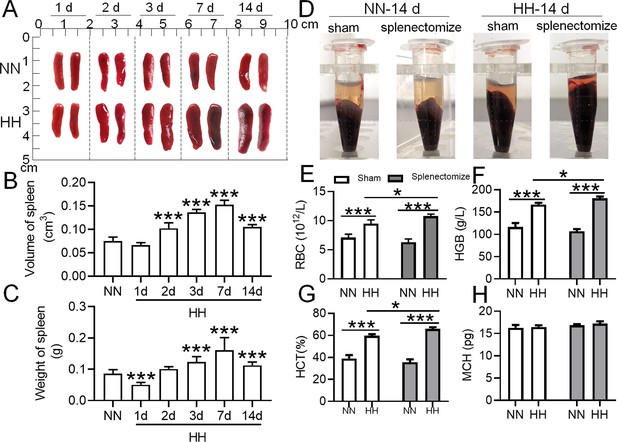
The spleen plays an important role in suppressing the immoderate increase in RBCs under HH conditions.
C57BL/6 mice with or without splenectomy were treated with NN and HH for varying durations, and the spleen and blood were collected for subsequent analyses. (A) Morphological observation, (B) Spleen volume, and (C) spleen weight was determined. (D–H) Blood observation and hematological index detection followed. Data are expressed as the means ± SEM (n=9 per group); * p<0.05, ** p<0.01, *** p<0.001 versus the NN group or the indicated group.
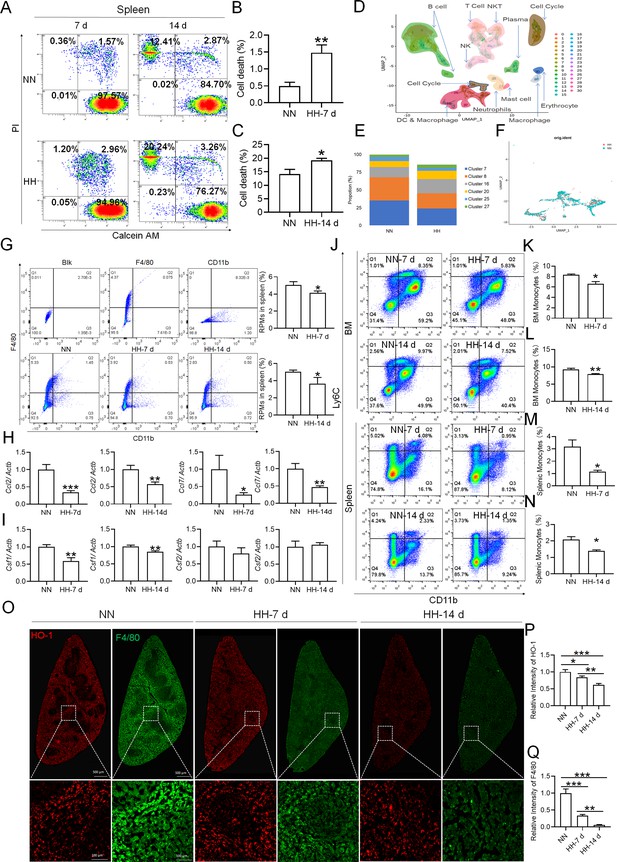
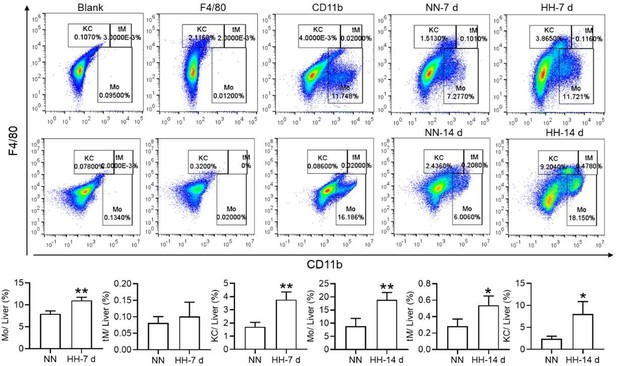
HH exposure results in a decrease in the number of splenic macrophages.
C57BL/6 mice were treated with NN and HH for 7 or 14 days, and the spleen and blood were collected for subsequent analysis. (A) Calcein/PI double staining was analysed by flow cytometry to assess cell viability. (B and C) The bar graphs represent the proportions of cell death in total splenic cell population. (D) Uniform Manifold Approximation and Projection (UMAP) provided a visualization of spleen cell clusters, with each color representing a unique cluster characterized by specific gene expression profiles. (E) Comparative analysis of the proportions of distinct macrophage clusters in spleens from NN- and HH-treated mice. (F) Presentation of individual macrophages from spleens of mice treated with either NN or HH. (G) Analysis of F4/80 and CD11b expression in splenic cells, conducted via flow cytometry. (H) qPCR analysis evaluated Ccl2 and Ccl7 gene expression in the spleen. (I) qPCR analysis of Csf1 and Csf2 expression levels in the spleen. (J) Flow cytometry facilitated the detection of CD11b and Ly6C double-stained cells in BM and spleen. (K–L) Bar graphs represent the proportions of CD11bhiLy6Chi cells in the BM, while (M–N) delineate those in the spleen. (O) HO-1 and F4/80 expression levels in the spleen were monitored after 0 (NN), 7, and 14 days of HH exposure. (P) The relative fluorescence intensities of HO-1 and (Q) F4/80, as outlined in (O), were quantitatively assessed. Data are expressed as means ± SEM for each group (n=3 per group). Statistical significance is indicated by * p<0.05, ** p<0.01, *** p<0.001 when compared to the NN group or the indicated group.
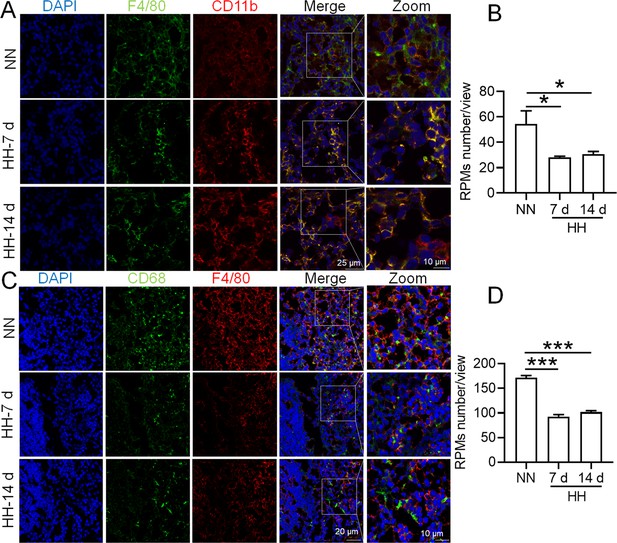
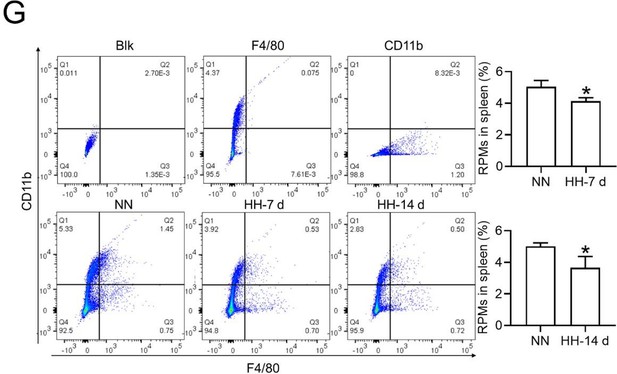
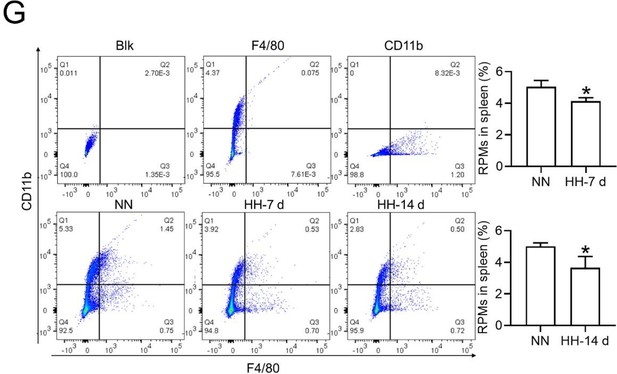
HH exposure induces RPMs reduction in the spleen.
(A) Immunofluorescence techniques were utilized to concurrently detect F4/80 and CD11b, and (C) F4/80 and CD68, to identify RPM contents in the spleen post-perfusion at 7 and 14 days of HH exposure. (B and D) Quantitative analyses of F4/80hiCD11blo and F4/80hiCD68hi cell populations in the spleen, as described in (A and C), were conducted. Data presented in this figure are expressed as means ± SEM for each group (n=3 per group). Statistical significance is indicated by * p<0.05, *** p<0.001 when compared to the NN group or as indicated. The results demonstrate a significant reduction in RPMs in the spleen following HH exposure, highlighting the effects of hypoxic conditions on splenic macrophage populations.

HH exposure induces transformation of RPMs subsets in the spleen.
This figure presents the analysis of RPMs in the spleen following 7 days of HH exposure, utilizing single-cell sequencing. (A) Marker genes were employed to identify RPMs within splenic cells, referencing Cell Marker 2.0. (B) RPMs in the spleen were categorized into three distinct clusters (0, 1, and 2) based on their gene expression profiles post-HH exposure. (C) Presenting the characteristic gene expression associated with each of these clusters in RPMs. (D) Pseudotime analysis of RPMs in the spleen was performed, with the trajectory figure illustrating the developmental pathway of these cells. This analysis elucidates the intricate changes in RPM subpopulations in the spleen under HH conditions, providing insights into the cellular dynamics and transcriptional alterations in response to hypoxia.
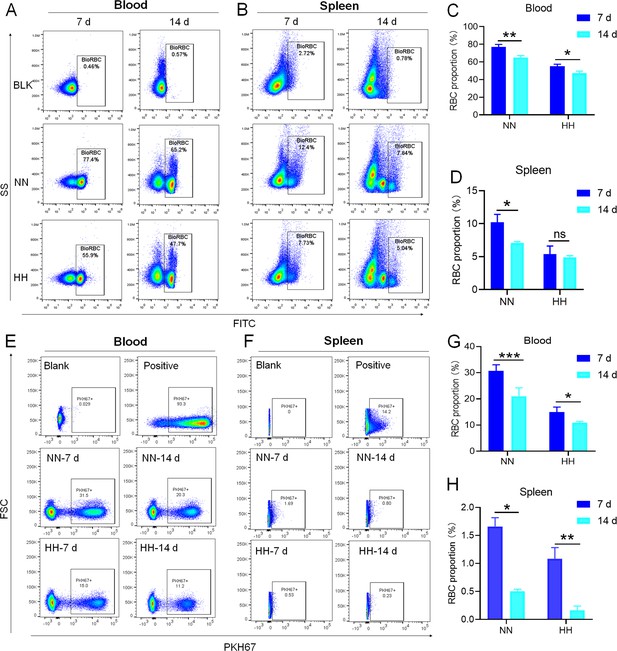
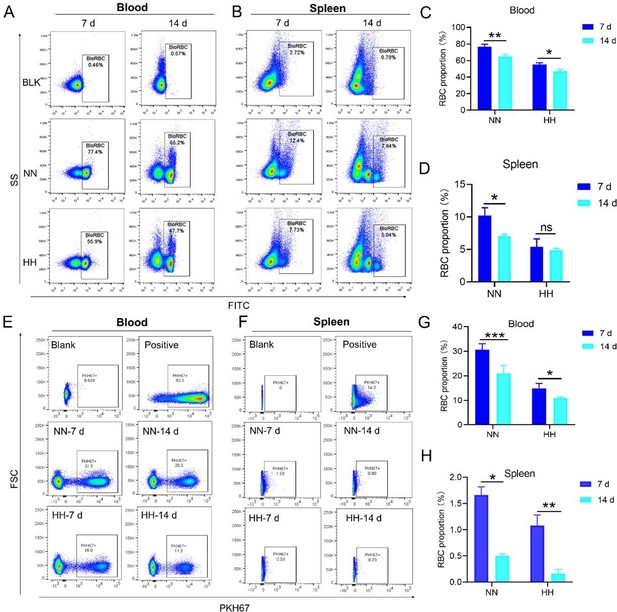
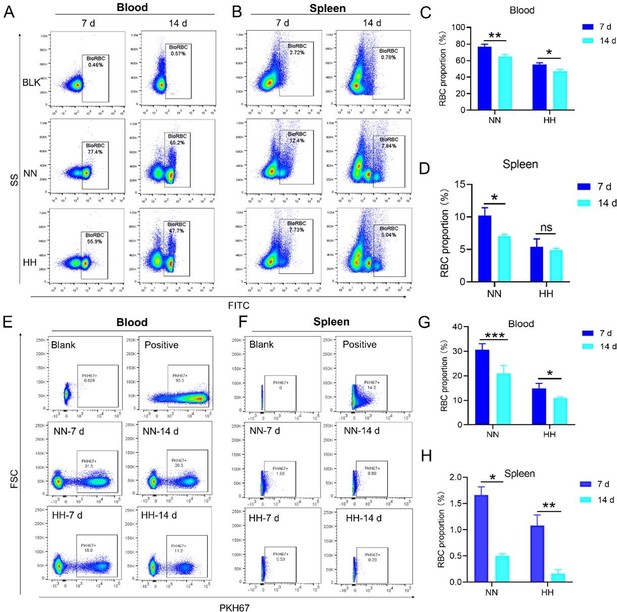
HH exposure decreases RBCs clearance both in the blood and spleen.
(A and B) Flow cytometry measurements illustrated the proportions of FITC-stained RBCs in blood and spleen, respectively, after 7 and 14 days of HH exposure. (C and D) Bar graphs depicted the quantified proportions of FITC-positive RBCs in both blood and spleen, respectively. Further, (E and F) presented flow cytometry assessments of the proportions of PKH67-labelled RBCs in blood and spleen following 7 and 14 days of HH exposure. The corresponding proportions of PKH67-positive RBCs in blood (G) and spleen (H) were also shown in bar graph format. The data are expressed as means ± SEM for each experimental group (n=3 per group). Statistical significance is denoted with * p<0.05, ** p<0.01, *** p<0.001, relative to the NN group or as specified in the graph legends. This compilation of data underlines the impact of HH exposure on the reduction of RBCs clearance in both blood and splenic compartments.
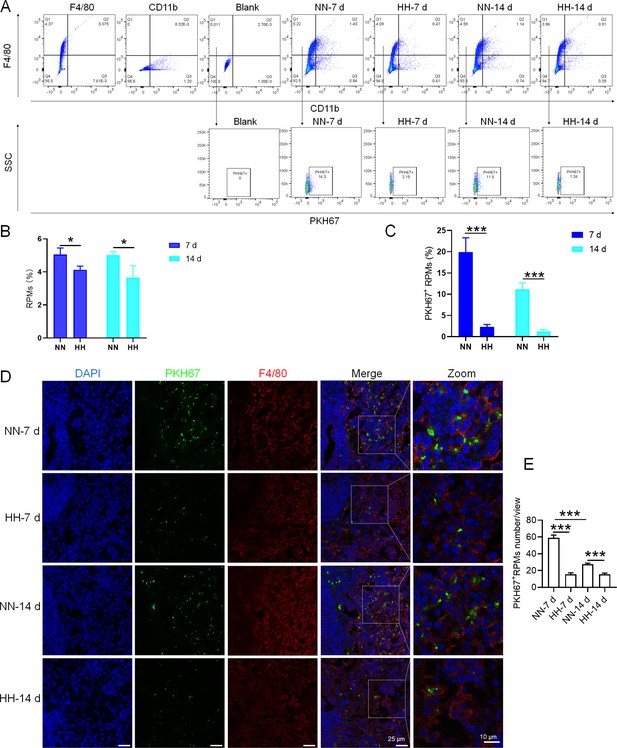
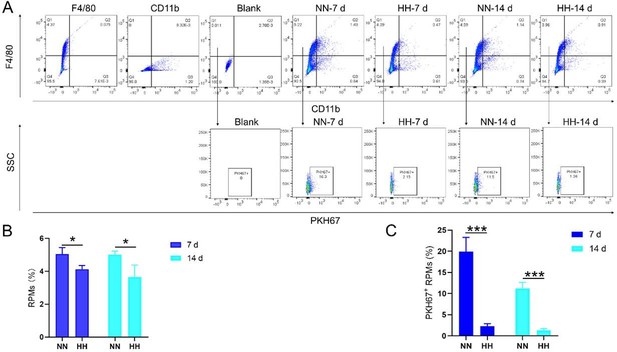
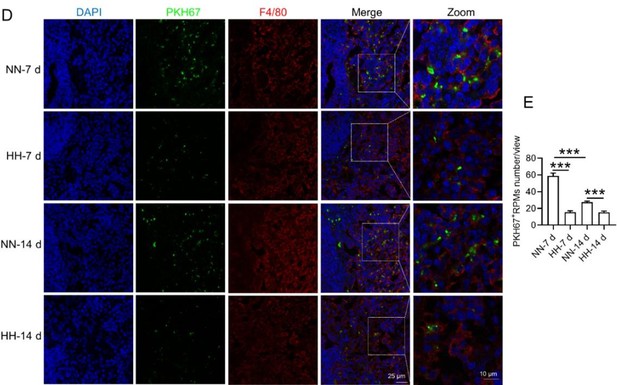
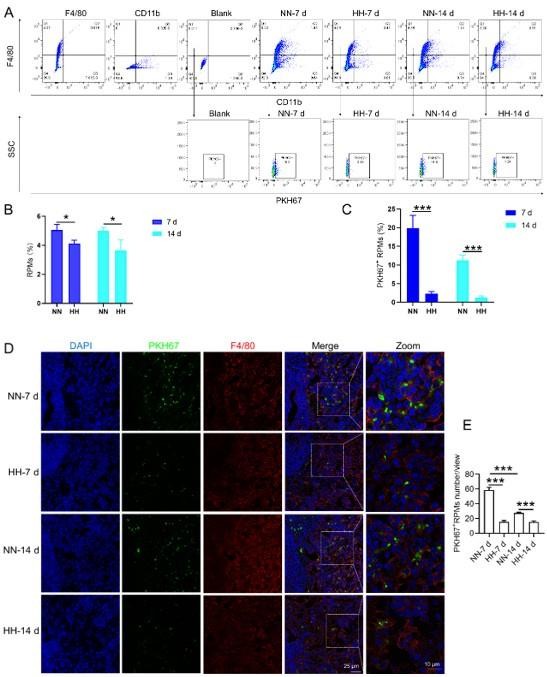
HH exposure reduces erythrophagocytosis in the spleen.
Mice were administered PKH67-labeled RBCs, followed by exposure to HH for durations of 7 and 14 days. (A) Flow cytometry was employed to analyze the proportions of RPMs (identified as F4/80+CD11b-) and PKH67-positive RPMs in the spleen post-exposure. (B and C) Bar graphs represent the quantified proportions of RPMs (F4/80+CD11b-) and PKH67-positive RPMs in the spleen, respectively. (D) The spleens were subjected to immunofluorescence analysis to detect F4/80 in conjunction with PKH67 fluorescence post-perfusion at 7 and 14 days following HH exposure. (E) Subsequent quantification of the number of PKH67-positive F4/80hi cells in the spleen, as depicted in (D), was conducted. The data derived from these analyses are expressed as means ± SEM for each group (n=3 per group). Statistical significance is denoted with * p<0.05, and *** p<0.001, relative to the indicated group in the graph legends.
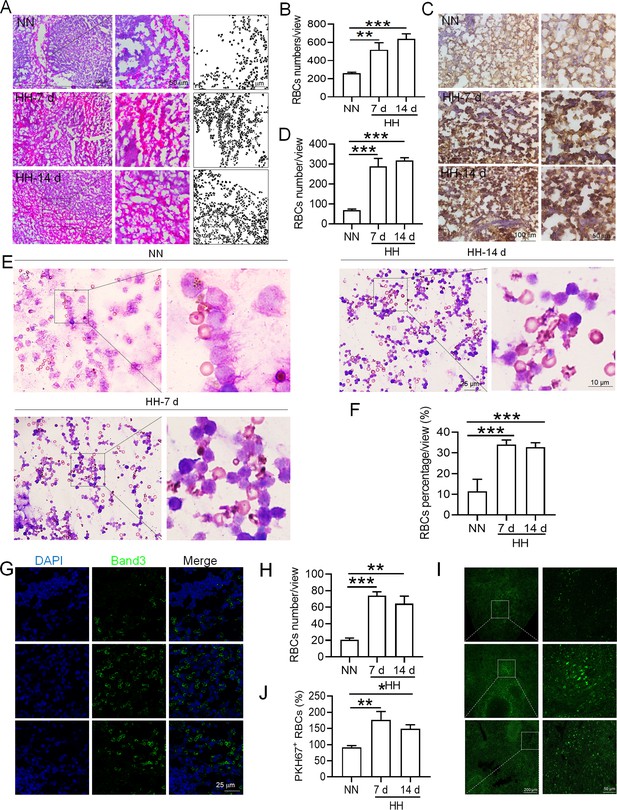
HH exposure increases RBCs retention in the spleen.
(A) HE staining was utilized to examine the presence of RBCs in the spleen post-perfusion at 7 and 14 days following HH exposure. (B) The number of RBCs present in the spleen, as observed in (A), was quantitatively assessed. (C and G) Immunohistochemical and immunofluorescent analyses using Band 3 staining were conducted to evaluate RBCs content in the spleen post-perfusion at 7 and 14 days of HH exposure. (D and H) The expression levels of Band 3 in the spleen, as shown in (C and G), were quantified. (E) Wright-Giemsa composite staining was performed on splenic cells following HH exposure for durations of 0 (NN), 7, and 14 days. (F) The proportion of RBCs within the perfused splenic cell population was determined. (I) Detection of PKH67 fluorescence in the spleen, without perfusion, was conducted after administering PKH67-labeled RBCs and subjecting them to HH exposure for 7 and 14 days. (J) The number of PKH67-positive RBCs within the spleen, as outlined in (I), was quantified. Data presented in this figure are expressed as means ± SEM for each experimental group (n=3 per group). Statistical significance is indicated by * p<0.05, ** p<0.01, *** p<0.001 when compared to the NN group or as specified.
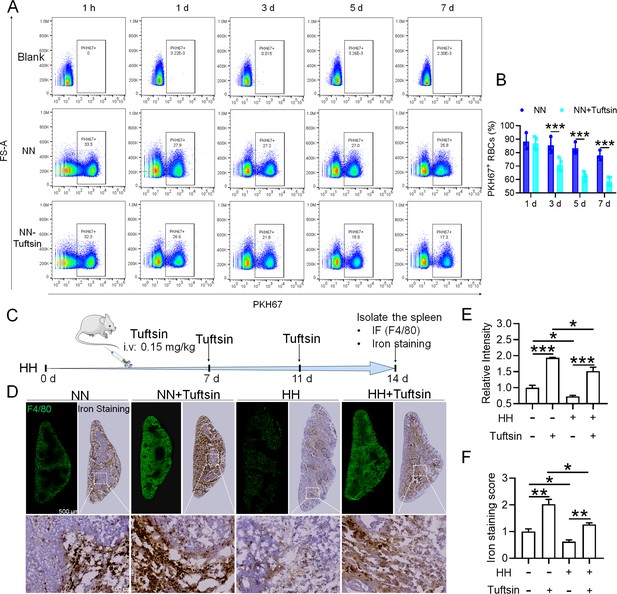
Impaired erythrophagocytosis of RBCs after HH exposure leads to a decrease in iron processing capacity in the spleen.
To stimulate phagocytosis of macrophages, Tuftsin was administered immediately following the injection of PKH67-labeled RBCs into mice. Subsequent to various exposure durations under NN conditions (1 hr, 1, 3, 5, or 7 days), (A) PKH67 fluorescence within the spleen was measured using flow cytometry. (B) The percentages of PKH67-positive RBCs in the spleen are represented in bar graph format. (C) The experimental design is depicted, wherein C57BL/6 mice (n=3 per group) were administered a single intravenous dose of Tuftsin (0.15 mg/kg) on days 7 and 11, followed by HH exposure until day 14, culminating in spleen isolation for F4/80 immunohistochemistry and iron staining. (D) Demonstrates F4/80 and iron staining in the spleen after 14 days of HH exposure with Tuftsin treatment. (E) Quantitative analysis of the relative fluorescence intensity of F4/80 expression in the spleen as described in (D). (F) Semi-quantitative assessment of iron levels in the spleen as outlined in (D). Data presented in this figure are articulated as means ± SEM for each group (n=3 per group). Statistical significance is denoted by * p<0.05, ** p<0.01, *** p<0.001 when compared to the NN group or as indicated.
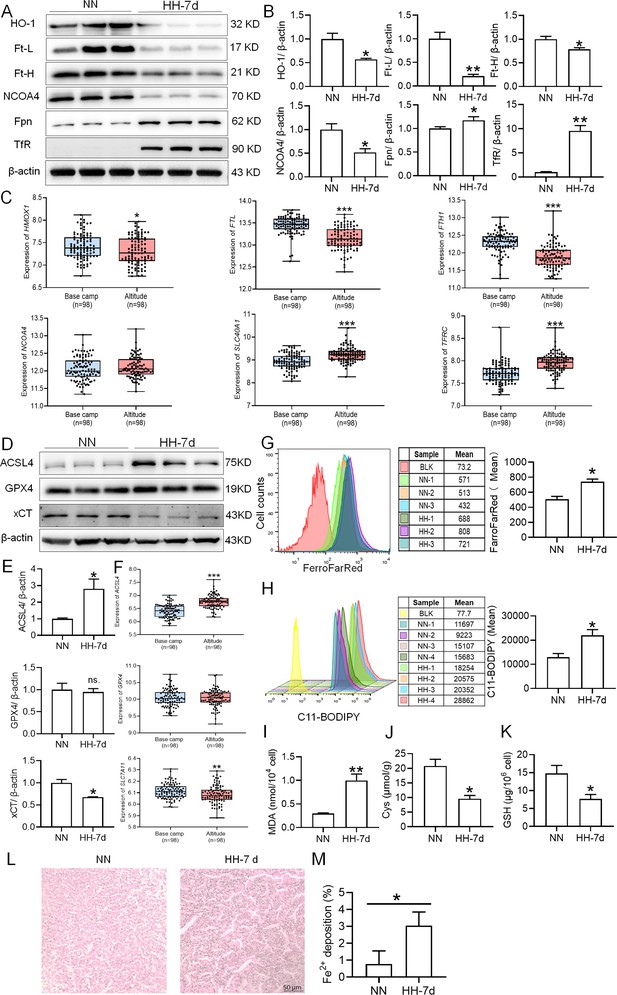
HH exposure enhances iron mobilization and induces ferroptosis in the spleen.
The C57BL/6 mice were treated with NN and HH for 7 days, and the spleen was collected for subsequent detection. (A) Western blot detection of HO-1, Ft-L, Ft-H, NCOA4, Fpn and TfR protein expression in spleen. (B) Statistical analysis of HO-1, Ft-L, Ft-H, NCOA4, Fpn, and TfR protein expression. (C) GEO data analysis of HMOX1, FTL, FTH1, NCOA4, SLC40A1, and TFRC mRNA expression in PMBCs before and after climbing to HA (n=98). (D) Western blot analysis for ACSL4, GPX4, and xCT protein expression in the spleen, with (E) depicting the statistical analysis of these protein levels. (F) GEO data analysis of ACSL4, GPX4 and SLC7A11 mRNA expression in PMBCs before and after reaching HA (n=98). (G) The content of Fe2+ in the spleen was detected using the FerroFarRed probe by flow cytometry. (H) The level of lipid ROS in the spleen was detected using the C11-BODIPY probe by flow cytometry. The MDA (I), Cys (J) and GSH (K) levels in the spleen were detected using biochemical detection kits, respectively. (L) Lillie staining of Fe2+ in the spleen after 7 days of HH exposure. (M) Fe2+ deposition in the spleen as described in (L) was quantified. Data are expressed as the means ± SEM (n=3 per group); * p<0.05, ** p<0.01, *** p<0.001 versus the NN group or the indicated group.
-
Figure 8—source data 1
PDF containing Figure 8A and original scans of the relevant western blot analysis (anti-HO-1, anti-Ft-L, anti-Ft-H, anti-NCOA4, anti-Fpn, anti-TfR and anti-β-actin) with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/87496/elife-87496-fig8-data1-v1.zip
-
Figure 8—source data 2
PDF containing Figure 8D and original scans of the relevant western blot analysis (anti-ACSL4, anti-GPX4, anti-xCT and anti-β-actin) with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/87496/elife-87496-fig8-data2-v1.zip

HH exposure promotes iron mobilization and induces ferroptosis in the spleen.
The C57BL/6 mice were treated with NN and HH for 14 days, and the spleen was collected for subsequent detection. (A) Western blot analysis was conducted to assess the expression levels of HO-1, Ft-L, Ft-H, NCOA4, Fpn, and TfR proteins in the spleen. (B) Statistical analysis of the protein expression levels detailed in (A). (C) Western blot analysis for ACSL4, GPX4, and xCT protein expression in the spleen. (D) The statistical analysis of these protein expression as shown in (C). (E) Flow cytometry using the FerroFarRed probe was employed to detect Fe2+ content in the spleen. (F) Quantitative results of the mean fluorescence intensity from (E). (G) Lipid ROS levels in the spleen were measured using the C11-BODIPY probe and flow cytometry. (H) Quantification of mean fluorescence intensity from (G). Levels of MDA (I), Cys (J), and GSH (K) in the spleen were determined using specific biochemical detection kits. (L) Lillie staining was performed to visualize Fe2+ in the spleen post 14 day HH exposure, with (M) providing a quantification of Fe2+ deposition as depicted in (L). Data are expressed as means ± SEM for each group (n=3 per group). Statistical significance is denoted by * p<0.05, ** p<0.01, *** p<0.001 when compared to the NN group or as indicated. These results collectively emphasize the significant impact of HH on the iron metabolism in the spleen, particularly highlighting the increased mobilization of iron and the onset of ferroptosis under hypoxic conditions.
-
Figure 8—figure supplement 1—source data 1
PDF containing Figure 8—figure supplement 1A and original scans of the relevant Western blot analysis (anti-HO-1, anti-Ft-L, anti-Ft-H, anti-NCOA4, anti-Fpn, anti-TfR and anti-β-actin) with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/87496/elife-87496-fig8-figsupp1-data1-v1.zip
-
Figure 8—figure supplement 1—source data 2
PDF containing Figure 8—figure supplement 1C and original scans of the relevant western blot analysis (anti-ACSL4, anti-Gpx4, anti-XCT, and anti-β-actin) with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/87496/elife-87496-fig8-figsupp1-data2-v1.zip
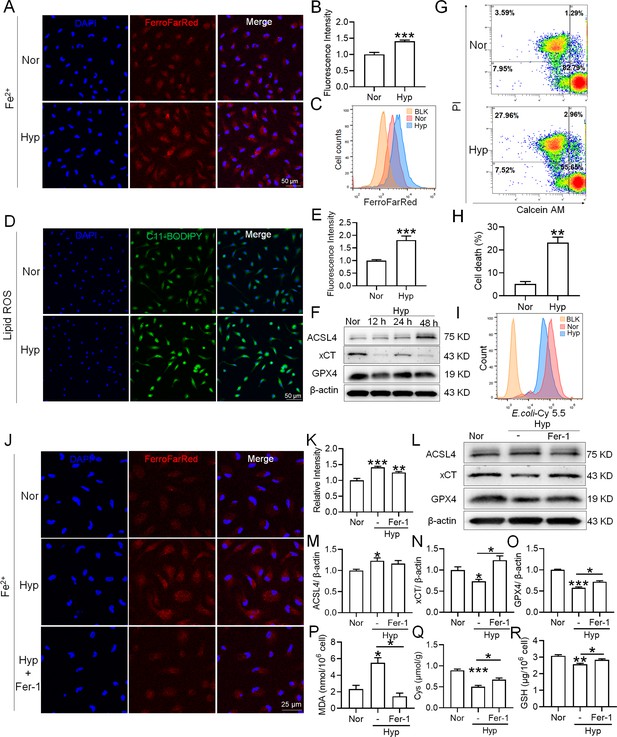
Hypoxia enhances the ferroptosis in splenic macrophage.
Splenic macrophages were cultured under 1% hypoxia for varying durations (0, 12, 24, 48 hr), either with or without a pre-treatment of 10 µM Fer-1 for 1 hr, before being collected for further examination. (A) Immunofluorescence detection of Fe2+ levels in macrophages after hypoxia exposure for 24 hr using the FerroFarRed probe under a fluorescence microscope. (B) Quantitative results of fluorescence intensity in the macrophages described in A. (C) Flow cytometry detection of Fe2+ levels in macrophages exposed to hypoxia for 24 hr using the FerroFarRed probe. (D) Immunofluorescence detection of lipid ROS levels in macrophages exposed to hypoxia for 24 hr using the C11-BODIPY probe under a fluorescence microscope. (E) Quantitative results of mean fluorescence intensity in the macrophages described in (D). (F) Western blot detection of ACSL4, GPX4, and xCT protein expression in macrophages under hypoxia for different times. (G) Flow cytometry was employed to analyze Calcein/PI double staining in macrophages following a 24 hr hypoxia exposure. (H) Cell death proportions in macrophages are given as bar graphs. (I) Macrophage phagocytic activity against Cy5.5-labelled E. coli following 24 hr of hypoxia exposure was assessed using flow cytometry in vitro. (J) Fe2+ levels in macrophages pre-treated with Fer-1, then exposed to 24 hr of hypoxia, were detected via the FerroFarRed probe in immunofluorescence. (K) Quantitative results of fluorescence intensity in the macrophages described in (J). (L) Western blot detection of ACSL4, GPX4, and xCT protein expression in macrophages pre-treated with Fer-1, then followed by 24 hr of hypoxia exposure. (M–O) Statistical analysis of ACSL4, xCT, and GPX4 protein expression in L. MDA (P), Cys (Q), and GSH (R) levels in macrophages pre-treated with Fer-1 and subsequently exposed to 24 hr of hypoxia were detected using kits and biochemical methods. Data are expressed as the means ± SEM (n=3 per group); * p<0.05, ** p<0.01, *** p<0.001 versus the NN group or the indicated group.
-
Figure 9—source data 1
PDF containing Figure 9F and original scans of the relevant western blot analysis (anti-ACSL4, anti-xCT, anti-GPX4, and anti-β-actin) with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/87496/elife-87496-fig9-data1-v1.zip
-
Figure 9—source data 2
PDF containing Figure 9L and original scans of the relevant western blot analysis (anti-ACSL4, anti-xCT, anti-GPX4, and anti-β-actin) with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/87496/elife-87496-fig9-data2-v1.zip
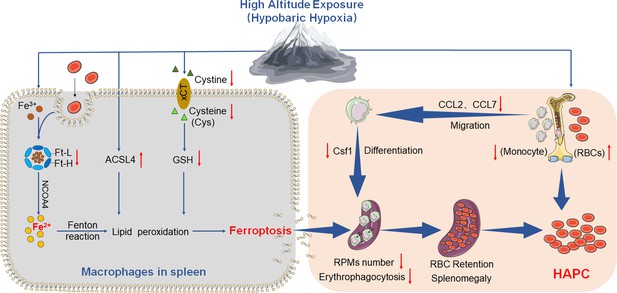
HA/HH exposure suppresses erythrophagocytosis by inducing macrophages ferroptosis in the spleen.
This figure summarizes the multifaceted impact of HA and HH exposure on splenic macrophage function. HA/HH exposure is demonstrated to trigger significant iron mobilization, notably increasing Fe2+ levels, which are further elevated in the spleen and macrophages of mice. Concurrently, HA/HH exposure is associated with an upregulation of ACSL4 expression, coupled with a suppression of xCT expression and GSH production. This alteration results in enhanced lipid peroxidation, culminating in the escalation of macrophage ferroptosis. Additionally, the exposure adversely affects monocyte migration and differentiation from BM to the spleen. Collectively, these effects of HA/HH exposure contribute to a notable inhibition of erythrophagocytosis, thereby promoting erythrocytosis and potentially exacerbating the progression of HAPC. This comprehensive analysis underscores the intricate interplay between environmental factors and cellular mechanisms in the spleen, which could significantly influence the pathogenesis of HAPC.