Functional partitioning of a liquid-like organelle during assembly of axonemal dyneins
Figures
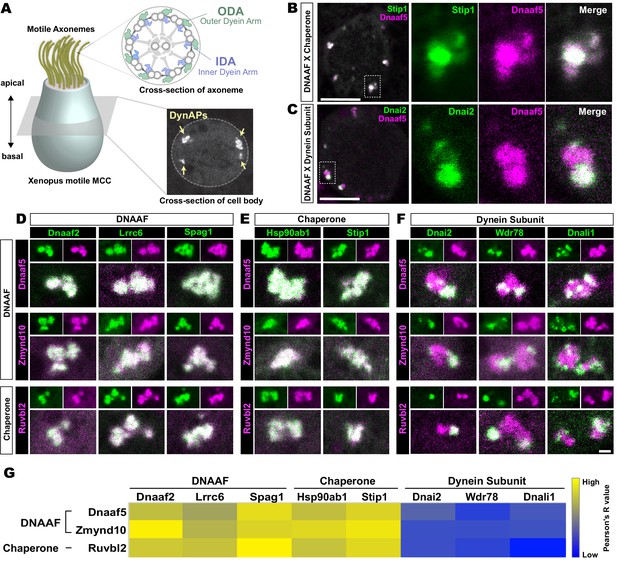
Dynein subunits occupy sub-regions within DynAPs.
(A) Schematic showing an MCC. The upper inset shows a schematic cross section through an axoneme indicating the relative positions of ODAs and IDAs; the lower inset shows a representative en face optical section through the MCC cytoplasm with dynAPs indicated DynAPs. (B) En face optical section through the cytoplasm of a Xenopus MCC expressing GFP-Stip1 (a chaperone, green) and mCherry-Dnaaf5 (a DNAAF, magenta). Note near-perfect co-localization. Dashed box indicates region shown in high magnification, split-channel views at right. Scale bars = 10 µm. (C) GFP-Dnai2 (a dynein subunit, green) co-localizes only partially with mCherry-Dnaaf5. (D, E) Images showing enlarged views of individual DynAPs after pairwise labeling with the indicated tagged DNAAFs or chaperones. Smaller panels show split channels, larger images show the merged channels. Note high degree of co-localization. (F) Similar pair-wise labeling with dynein subunits reveals only partial co-localization. Scale bar = 1 µm for panels D-F. (G) Heatmap showing Pearson correlation for colocalization of GFP fusion proteins with mCherry fusion proteins. Yellow indicates high value (Pearson’s R value = 0.85), while blue indicates low value (Pearson’s R value = 0.45). Exact Pearson’s R values for each protein combination can be found in Figure 1—figure supplement 1.
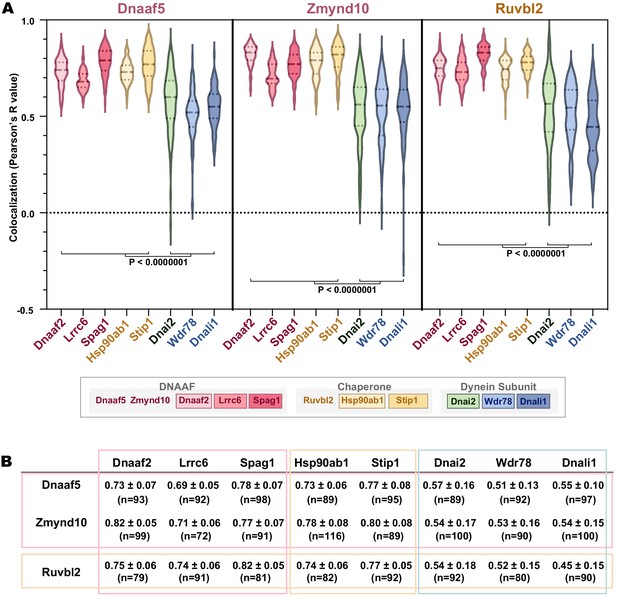
Quantification of localization data.
(A) Graph displaying Pearson correlation for colocalizations between DNAAF, chaperone or dynein subunits. Dnaaf5, Zmynd10 and Ruvbl2 were tagged with mCherry. GFP fusion proteins are Dnaaf2, Lrrc6, Spag1, Hsp90ab1, Stip1, Dnai2, Wdr78 and Dnali1. p-value<0.0000001 by one-way ANOVA and post-hoc Tukey-Kramer HSD test. (B) Table showing Pearson’s R values (Average ± STDEV) and n-value for each colocalization analysis (at least 15 cells from five embryos for each analysis).
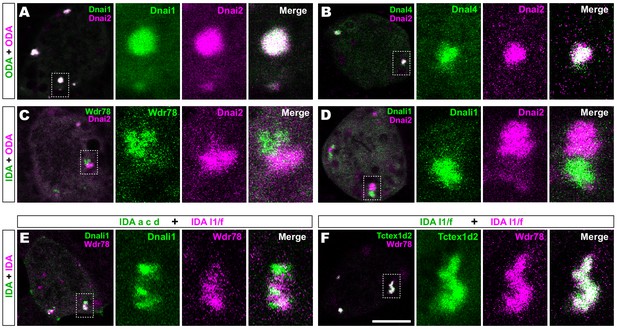
Outer and inner dynein arms localize to distinct sub-regions in DynAPs.
(A) En face optical section of a Xenopus MCC expressing ODA subunits GFP-Dnai1 and mCherry-Dnai2 (as per lower inset in Figure 1A). The dashed box indicates the area shown at higher magnification in the panels to the right. Note near complete co-localization. (B) mCherry-Dnai2 also displays near-total co-localization with another ODA subunit, Dnal4. (C, D) mCherry-Dnai2 shows very little co-localization with the IDA subunit GFP-Wdr78 or GFP-Dnali1. (E) IDA-f subunit mCherry-Wdr78 displays little co-localization with IDA-a, c, d subunit GFP-Dnali1. (F) IDA-f subunits, mCherry-Wdr78 and GFP-Tctex1d2, show complete co-localization. Scale bar = 10 µm.
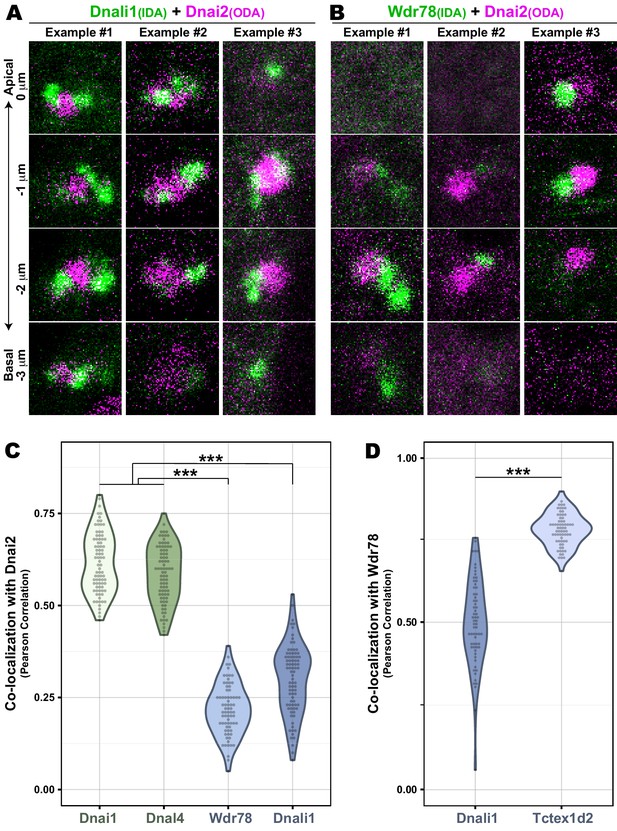
Co-localization data for IDAs and ODAs in DynAPs.
(A) En face optical Z-sections through DynAPs possessing GFP-Dnali1 and mCherry-Dnai2. (B) En face optical Z-sections through DynAPs possessing GFP-Wdr78 and mCherry-Dnai2. (C) Graph displaying Pearson correlation for colocalizations of GFP fusion dynein subunits, Dnai1 (n = 82), Dnal4 (n = 83), Wdr78 (n = 73) and Dnali1 (n = 83), with mCherry-Dnai2 at DynAPs. p-value (***)<0.0000001 by one-way ANOVA and post-hoc Tukey-Kramer HSD test. (D) Graph displaying Pearson correlation for colocalization of two different IDA subunits, GFP-Dnali1 (n = 65) and GFP-Tctex1d2 (n = 67) with mCherry-Wdr78 at DynAPs. p-value (***)<2.2e-16 by Welch Two Sample t-test.
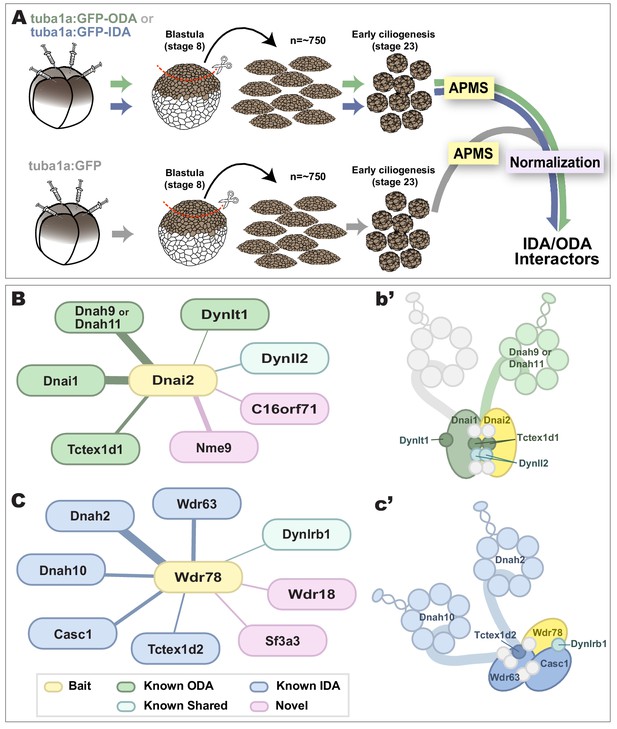
Specific identification of outer and inner arm dynein interactors.
(A) Schematic of APMS workflow for identifying in vivo ODA and IDA interactors. GFP-tagged ODA or IDA driven by MCC-specific alpha-tubulin promoter by plasmid injection into Xenopus embryos and animal caps were isolated at stage 8. The cultured explants were collected at early ciliogenesis stage (stage 23) and subjected to APMS. Unfused GFP was assessed simultaneously and the data subtracted to control for non-specific interactions. (B) Spoke diagram displaying Dnai2 (ODA) interactors, line weight of spokes indicates Log2 fold-change of PSMs. b’. Schematic of outer dynein arm indicating identified Dnai2 preys. (C) Spoke diagram displaying Wdr78 (IDA) interactors. c’. Schematic of inner dynein arm (f type) indicating identified Wdr78 preys.
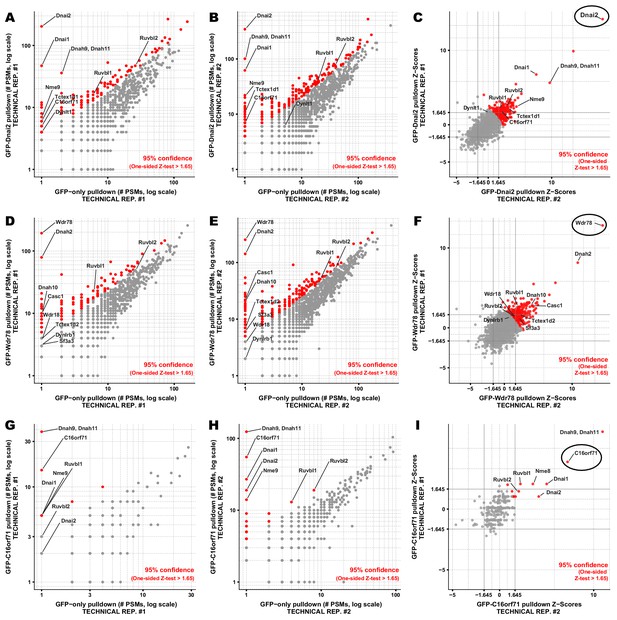
Proteins co-precipitating with Dnai2, Wdr78, and C16orf71 by APMS.
Interaction partners of (A-C) Dnai2, (D-F) Wdr78, and (G-I) C16orf71 were identified based on their enrichment in APMS of the GFP-tagged bait proteins (vertical axes, panels A-B, D-E, G-H) relative to APMS of the untagged GFP controls (horizontal axes, panels A-B, D-E, G-H). A pseudocount of 1 PSM was added to each protein for visualization on a log-log plot (panels A-B, D-E, G-H). For visualization purposes, each protein was colored according to a 95% confidence threshold using a one-sided Z-test (see Materials and methods). We observed reasonable concordance between technical replicates, evident in comparisons of the measured Z-scores for proteins co-precipitating with (C) Dnai2, (F) Wdr78, and (I) C16orf71. Baits are circled in C, F, I. A joint Z-score was calculated for each protein observed across a pair of technical replicates by summing the Z-score for each protein in each replicate and dividing by the square root of N replicates (N = 2). Multiple-hypothesis corrected p-values derived from these joint Z-scores are provided in the corresponding Suppl. Tables.
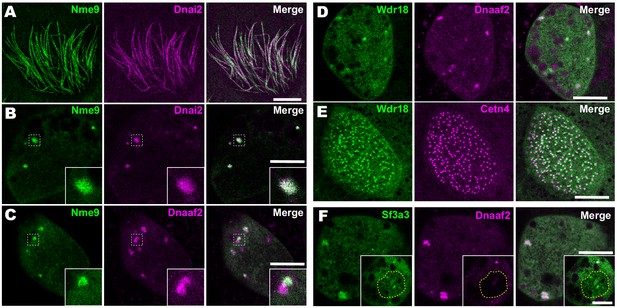
Localization of novel IDA and ODA interactors.
(A) En face optical section just above the apical surface of a Xenopus MCC showing axonemal localization of GFP-Nme9. (B) A similar optical section through the MCC cytoplasm shows near-perfect colocalization of GFP-Nme9 with the ODA subunit mCherry-Dnai2 in DynAPs. Dashed box indicates area shown at higher magnification in the inset. (C) GFP-Nme9 only partially co-localizes with mCherry-Dnaaf2. (D) En face optical section through the cytoplasm shows colocalization of GFP-Wdr18 with mCherry-Dnaaf2 in DynAPs. (E) A similar section just below the MCC apical surface reveals GFP-Wdr18 localization near basal bodies labeled with RFP-Centrin4 (F) GFP-Sf3a3 co-localizes with mCherry-Dnaaf2. Scale bars = 10 µm.
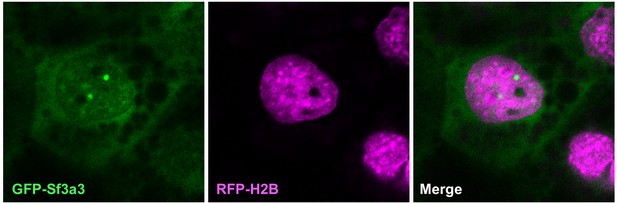
Sf3a3-GFP (green) also localizes to speckles within the nucleus (labeled with RFP-Histone2B, red) in a Xenopus MCC.
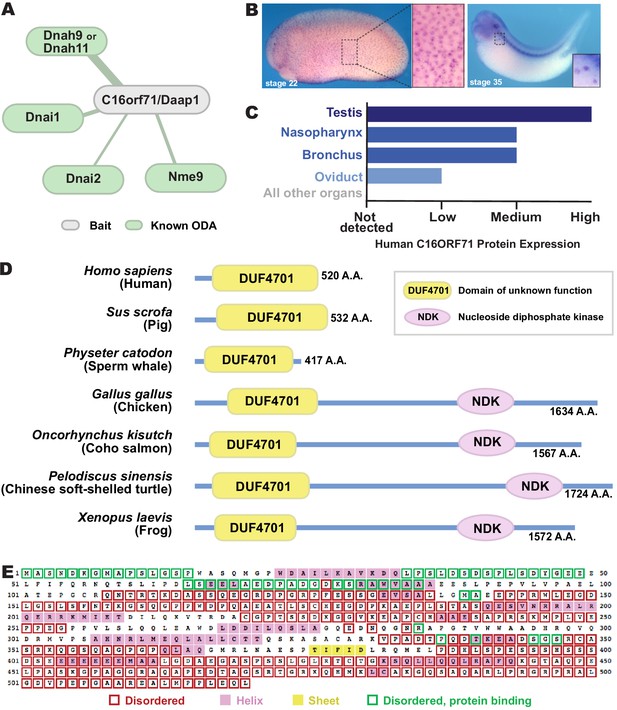
C16orf71/Daap1 is a novel ODA interactor specifically expressed in MCCs.
(A) Spoke diagram displaying C16orf71/Daap1 interactors; line weight indicates Log2 fold- change of PSMs. (B) In situ hybridization of Daap1 in Xenopus reveals expression in epidermal MCCs (left) and MCCs in the nephrostomes (right). (C) Graph showing C16orf71 protein expression levels in human tissues from the Human Protein Atlas (Uhlén et al., 2015). (D) Domain organization of C16orf71/Daap1 orthologs across vertebrates from NCBI Orthologs. (E) Domain prediction with human C16orf71/DAAP1 sequence from the PSIPRED Protein Analysis Workbench for disorder and secondary structure prediction.
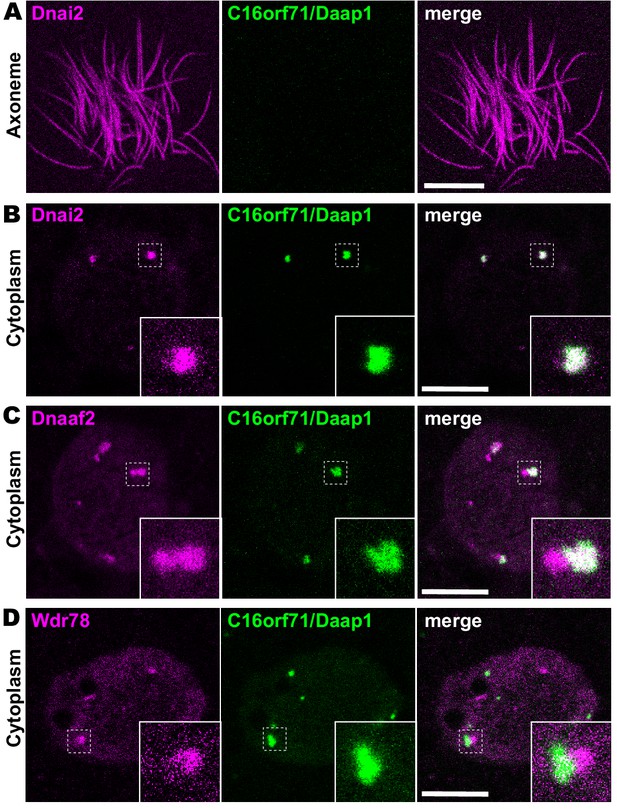
Daap1 is a DynAP-specific protein in Xenopus MCCs.
(A) En face optical section above the apical surface of a Xenopus MCC reveals absence of GFP-Daap1 in axonemes. (B) A similar optical section through the MCC cytoplasm shows near-perfect colocalization of GFP-Daap1 with the ODA subunit mCherry-Dnai2 in DynAPs. Inset shows higher magnification view of dashed box for each panel. (C) GFP-Daap1 only partially co-localizes with mCherry-Dnaaf2. (D) GFP-Daap1 co-localizes only very weakly with the IDA subunit mCherry-Wdr78. Scale bars = 10 µm.
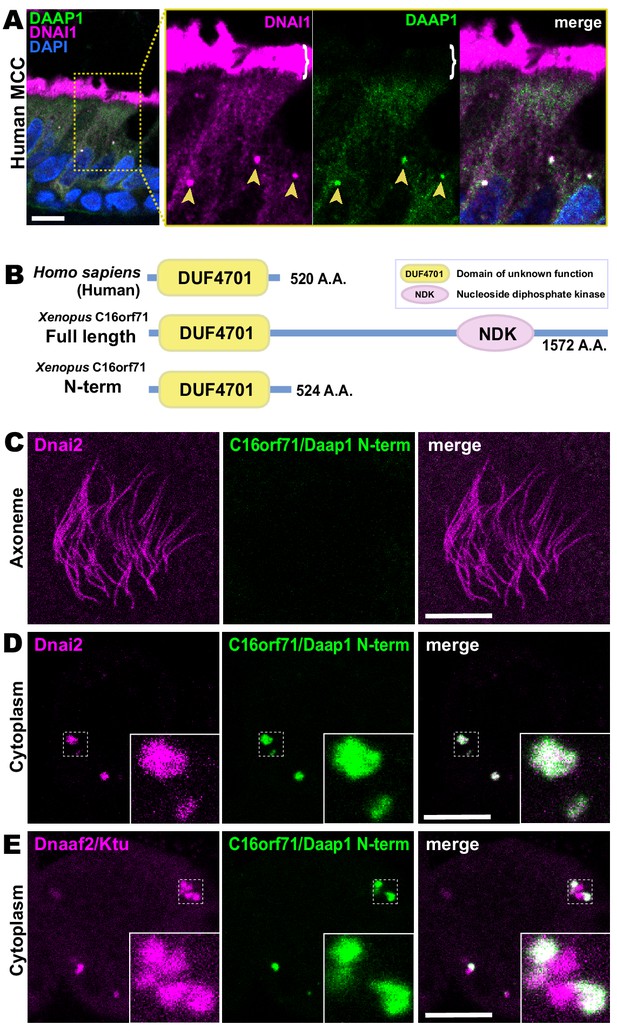
Human DAAP1 is a DynAP-specific protein.
(A) A primary human lung section immunostained for human DAAP1 (green) and the ODA subunit DNAI1 (magenta). Boxed area indicates magnified region shown at right. DNAI1 strongly labels MCC axonemes (bracket) and also DynAPs in the cytoplasm (arrowheads). As in Xenopus, human DAAP1 is not present in axonemes but is strongly enriched in DynAPs. (DAPI (blue) marks nuclei; scale bar = 10 µm) (B) Schematic of C16orf71 constructs: Xenopus Daap1-Nterm is truncated, containing only the DUF4701 domain, similar to human DAAP1. (C) En face optical section above the apical surface of a Xenopus MCC reveals absence of Daap1-Nterm-GFP in axonemes. (D, E) Similar optical sections through the cytoplasm shows near-perfect colocalization of GFP-Daap1-Nterm with the ODA subunit mCherry-Dnai2 and only partial co-localization with Dnaaf2/Ktu. In all cases, inset shows higher magnification view of dashed box for accompanying panels. Scale bars = 10 µm.
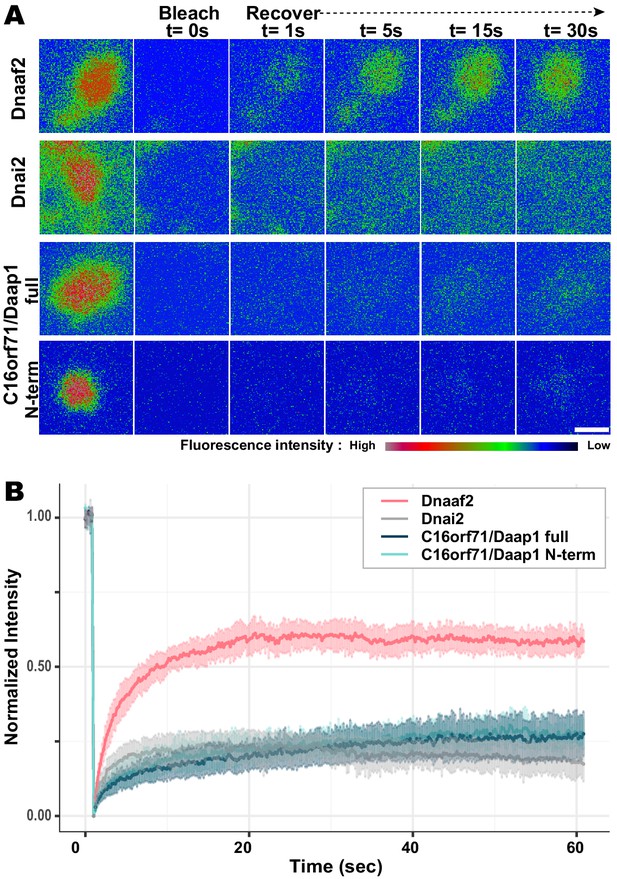
Daap1 is stably retained in DynAPs.
(A) Stills from time-lapse imaging of FRAP for the assembly factor GFP-Dnaaf2, the ODA subunit GFP-Dnai2, GFP-Daap1 and GFP-Daap1-Nterm. Images are color-coded to highlight changes in pixel intensity (see key below images). Scale bar = 1 µm (B) Graphs displaying FRAP curves for GFP-Dnaaf2 (n = 14), GFP-Dnai2 (n = 12) GFP-Daap1 (n = 26), and GFP-Daap1-Nterm (n = 22).
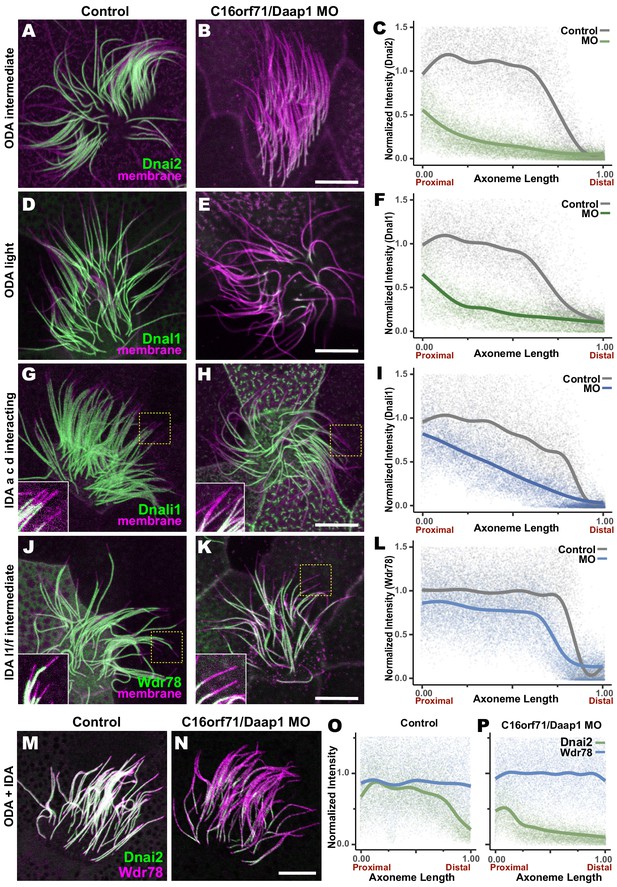
Loss of C16orf71 disrupts deployment of dynein subunits to the axoneme.
(A–B) Xenopus MCC axonemes labeled by membrane-RFP (magenta) together with the GFP-Dnai2, an ODA intermediated chain in control embryo (A) and in C16orf71/Daap1 morphant (B). (C) Graph showing intensity of GFP-Dnai2 along the normalized axoneme length (n = 29 for control, 51 for morphant). (D–E) MCCs labeled by membrane-RFP (magenta) together with the GFP-Dnal1, an ODA light chain in control embryo (D) and in C16orf71/Daap1 morphant (E). (F) Graph showing intensity of GFP-Dnal1 along the normalized axoneme length (n = 27 for control, 26 for morphant). (G–H) MCCs labeled by membrane RFP (magenta) together with the GFP-Dnali1, an IDA a, c, d interacting protein in control embryo (G) and in C16orf71/Daap1 morphant (H). (I) Graph showing intensity of GFP-Dnali1 along the normalized axoneme length (n = 30 for control, 33 for morphant). (J–K) MCCs labeled by membrane-RFP (magenta) together with the GFP-Wdr78, an IDA l1/f intermediated chain in control embryo (J) and in C16orf71/Daap1 morphant (K). (L) Graph showing intensity of GFP-Dnal1 along the normalized axoneme length (n = 32 for control, 43 for morphant). Both Dnai2, an ODA Intermediated chain and Dnal1, an ODA light chain are severely reduced in the axoneme in C16orf71 morphants, while IDA subunits display only mild loss from the distal most axoneme (inset). Yellow boxes indicate regions shown in accompanying insets for each panel. (M–N) MCCs co-expressing a marker for both ODAs (Dnai2, green) and IDAs (Wdr78, magenta). Loss of C16orf71 results in specific loss of ODAs in the axoneme (N), while both ODA and IDA properly localize in the axoneme in control (M). (O–P) Graphs showing intensities of GFP-Dnai2 and mCherry-Wdr78 along the normalized axoneme length in controls (O) and morphants (P). Scale bars = 10 µm.
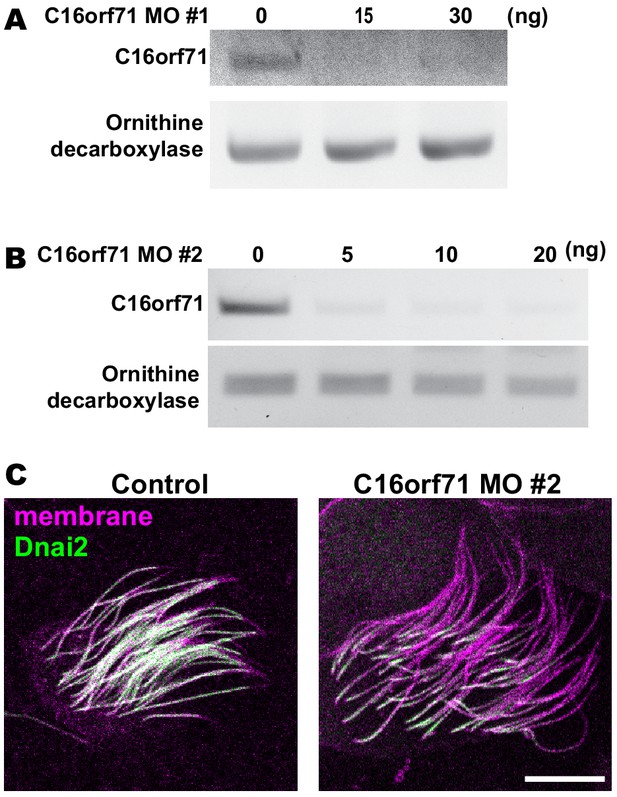
Validation of C16orf71 morpholinos.
(A–B) RT-PCR shows effective disruption of splicing by injection of C16orf71 MO #1 (A) and #2 (B). Ornithine decarboxylase (ODC) was used as a loading control for the RT-PCR. (C) Apical surface views labeled by membrane-RFP (magenta) and GFP-Dnai2 (green). Loss of C16orf71 by MO #2 results in reduction of Dnai2 in the axoneme, while GFP-Dnai2 reveals normal localization in control. Scale bar = 10 µm.
Additional files
-
Supplementary file 1
Table showing orthogroups and proteins with PSMs identified by APMS with Dnai2.
- https://cdn.elifesciences.org/articles/58662/elife-58662-supp1-v2.csv
-
Supplementary file 2
Table showing orthogroups and proteins with PSMs identified by APMS with Wdr78.
- https://cdn.elifesciences.org/articles/58662/elife-58662-supp2-v2.csv
-
Supplementary file 3
Table showing orthogroups and proteins with PSMs identified by APMS with Dnai2.
- https://cdn.elifesciences.org/articles/58662/elife-58662-supp3-v2.csv
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/58662/elife-58662-transrepform-v2.docx





