Alternative Splicing: A new role for Hif-1α
Animals have evolved complex respiratory systems which efficiently deliver oxygen to every part of their body. Still, bursts of oxygen shortages can occasionally take place when metabolic needs surpass supply. Long-distance flying, for example, requires desert locusts to consume 30–150 times more oxygen than they do at rest (Armstrong and Mordue, 1985).
In response, cells can recruit hypoxia-inducible factors – or Hif, for short – which are formed of combinations of Hif-α and Hif-ß modules. These highly conserved proteins can bind to genetic sequences (known as hypoxia-response elements) to regulate genes that control how cells and organisms adjust to a lack of oxygen (Schofield and Ratcliffe, 2004). Under normal conditions, Hif proteins undergo oxygen-dependent chemical modifications that lead to their degradation (Ivan et al., 2001; Min et al., 2002). Low levels of oxygen inhibit this process, stabilising the proteins and activating various hypoxia-related genes (Wenger et al., 2005).
Overall, regulating the activity of Hif proteins – and especially which genes they target – involves a wide range of pathways, molecules, and binding partners (Chandel et al., 2000; Guzy et al., 2005). These processes can even, on occasion, be independent of oxygen levels, allowing Hif proteins to participate in other types of life processes, such as the development of insect blood cells (Mukherjee et al., 2011).
In addition, several versions of the protein can exist within an organism. Some emerge from closely related Hif genes, but others are isoforms, being created from the same gene through various mechanisms. Now, in eLife, Le Kang, Bing Chen and colleagues at the Chinese Academy of Science and Hebei University – including Ding Ding as first author – report how an isoform of the Hif-1α gene contributes to the integrity and performance of insect muscles during flight (Ding et al., 2022).
The team focused on migratory locusts (Locusta migratoria), an agricultural pest that can fly hundreds of kilometers per day. Like all invertebrates, these insects express only one Hif-α gene, Hif-1α. Examining the entire coding sequence of this gene revealed that it leads to the production of two distinct isoforms (Hif-1α1 and Hif-1α2) via a process known as alternative splicing. This mechanism involves the cells reshuffling the coding elements present in the RNA transcripts of the Hif-1α gene, resulting in different proteins.
Ding et al. showed that in contrast to Hif-1α1 (which is only detectable under hypoxic conditions), Hif-1α2 lacks the domain required to respond to oxygen levels (Figure 1A). The isoform is abundantly expressed in flight muscles, regardless of oxygen concentration. Interestingly, the experiments showed that Hif-1α2 is essential for the prolonged flight performance of locusts, whereas Hif-1α1 has no effect on this trait.
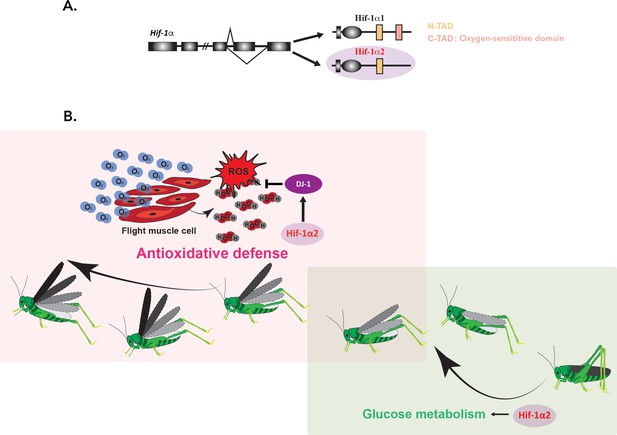
Hif-1α2 is required for long-term flight in migratory locusts.
(A) In migratory locusts, Hif-1α produces two distinct isoforms, Hif-1α1 and Hif-1α2 (lilac). Examining the genetic sequence of the two isoforms showed that, unlike Hif-1α1, Hif-1α2 does not carry the oxygen-sensitive C-TAD domain (peach) and it is therefore oxygen-insensitive. Hif-1α2 is expressed specifically in flight muscles. (B) During the initial stage of a flight (right; soft green), glucose metabolism is required to meet the need for high energy demands. Hif-1α2 (lilac) upregulates genes involved in this process, but its removal did not impact long-term flight behavior. As the insects continue to fly (left; soft pink), increased oxygen consumption (blue O2 molecules) and energy generation lead to an accumulation of harmful reactive oxygen species (ROS) such as hydrogen peroxide (H2O2; in red and grey). Hif-1α2 helps to deal with these dangerous metabolic byproducts by promoting the expression of the ‘ROS quencher’ DJ-1 (purple), which participates in the antioxidative defense. Animals lacking Hif-1α2 or DJ-1 therefore accumulate ROS in their flight muscles and are unable to fly for an extended period.
The team then genetically manipulated locusts to silence the Hif-1α2 isoform, before examining gene expression profiles in these mutants and in normal insects. This highlighted 12 genes that were significantly downregulated when the isoform was knocked down. Amongst these, 11 are involved in the same energy-creating pathway; however, interfering with this molecular cascade had no discernible effects on the insects’ ability to perform long-term flights. As a result, Ding et al. propose that the pathway plays a role earlier on, as the locust takes off and starts to fly.
The remaining gene, which codes for the DJ-1 protein, helps to protect the organism against reactive oxygen species (or ROS). This class of harmful molecules is released by cellular activity. In flight muscles, their constant presence is correlated with high oxygen consumption, and increases when Hif-1α2 is knocked down. In addition, Ding et al. showed that DJ-1 is enriched in flight muscles; if removed, flight-induced ROS levels soar up and performance becomes impaired.
Finally, the team confirmed a new, non-canonical target for Hif-1α2 by showing that the isoform binds to the hypoxia-response elements present in the promoter of the DJ-1 gene, even when oxygen levels are normal (Figure 1B). Overall, Ding et al. propose that Hif-1α2 confers a physical advantage in prolonged flight by alleviating the damage linked to ROS while simultaneously maintaining efficient energy production during the initial stage of flight.
Insects were among the first animals to fly, but while some have evolved to be one the most efficient creatures to have taken to the skies, others are far less skilled. The oxygen-sensitive domain of Hif-1α has been under high selective pressure throughout evolution; the presence of a range of isoforms for this gene, including oxygen-insensitive variants, is likely to contribute to this divergence in flight performance (Graham and Presnell, 2017). Future studies should investigate exactly how Hif-1α2 is alternatively spliced in flight muscles and how it is controlled independently of oxygen, both in locusts and in other insects. A better grasp of the remarkable versatility of the Hif pathway, including in humans, could help to pinpoint the evolutionary and ecological significance of these genes, and why certain Hif proteins are involved in developmental conditions or cancer.
References
-
Oxygen consumption of flying locustsPhysiological Entomology 10:353–358.https://doi.org/10.1111/j.1365-3032.1985.tb00057.x
-
Reactive oxygen species generated at mitochondrial complex III stabilize hypoxia-inducible factor-1alpha during hypoxia: a mechanism of O2 sensingThe Journal of Biological Chemistry 275:25130–25138.https://doi.org/10.1074/jbc.M001914200
-
Oxygen sensing by HIF hydroxylasesNature Reviews. Molecular Cell Biology 5:343–354.https://doi.org/10.1038/nrm1366
-
Integration of oxygen signaling at the consensus HREScience’s STKE 2005:re12.https://doi.org/10.1126/stke.3062005re12
Article and author information
Author details
Publication history
- Version of Record published: August 30, 2022 (version 1)
Copyright
© 2022, Shin and Shim
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 767
- views
-
- 175
- downloads
-
- 0
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Biochemistry and Chemical Biology
- Cell Biology
Hibernation is a period of metabolic suppression utilized by many small and large mammal species to survive during winter periods. As the underlying cellular and molecular mechanisms remain incompletely understood, our study aimed to determine whether skeletal muscle myosin and its metabolic efficiency undergo alterations during hibernation to optimize energy utilization. We isolated muscle fibers from small hibernators, Ictidomys tridecemlineatus and Eliomys quercinus and larger hibernators, Ursus arctos and Ursus americanus. We then conducted loaded Mant-ATP chase experiments alongside X-ray diffraction to measure resting myosin dynamics and its ATP demand. In parallel, we performed multiple proteomics analyses. Our results showed a preservation of myosin structure in U. arctos and U. americanus during hibernation, whilst in I. tridecemlineatus and E. quercinus, changes in myosin metabolic states during torpor unexpectedly led to higher levels in energy expenditure of type II, fast-twitch muscle fibers at ambient lab temperatures (20 °C). Upon repeating loaded Mant-ATP chase experiments at 8 °C (near the body temperature of torpid animals), we found that myosin ATP consumption in type II muscle fibers was reduced by 77–107% during torpor compared to active periods. Additionally, we observed Myh2 hyper-phosphorylation during torpor in I. tridecemilineatus, which was predicted to stabilize the myosin molecule. This may act as a potential molecular mechanism mitigating myosin-associated increases in skeletal muscle energy expenditure during periods of torpor in response to cold exposure. Altogether, we demonstrate that resting myosin is altered in hibernating mammals, contributing to significant changes to the ATP consumption of skeletal muscle. Additionally, we observe that it is further altered in response to cold exposure and highlight myosin as a potentially contributor to skeletal muscle non-shivering thermogenesis.
-
- Cell Biology
Quiescence (G0) maintenance and exit are crucial for tissue homeostasis and regeneration in mammals. Here, we show that methyl-CpG binding protein 2 (Mecp2) expression is cell cycle-dependent and negatively regulates quiescence exit in cultured cells and in an injury-induced liver regeneration mouse model. Specifically, acute reduction of Mecp2 is required for efficient quiescence exit as deletion of Mecp2 accelerates, while overexpression of Mecp2 delays quiescence exit, and forced expression of Mecp2 after Mecp2 conditional knockout rescues cell cycle reentry. The E3 ligase Nedd4 mediates the ubiquitination and degradation of Mecp2, and thus facilitates quiescence exit. A genome-wide study uncovered the dual role of Mecp2 in preventing quiescence exit by transcriptionally activating metabolic genes while repressing proliferation-associated genes. Particularly disruption of two nuclear receptors, Rara or Nr1h3, accelerates quiescence exit, mimicking the Mecp2 depletion phenotype. Our studies unravel a previously unrecognized role for Mecp2 as an essential regulator of quiescence exit and tissue regeneration.






