Exosomes: How tumors escape the immune system
All cells in the body release small nano-sized vesicles known as exosomes into the space around them. These vesicles contain a diverse array of biological materials, such as nucleic acids, proteins, lipids and other metabolites that represent the ‘parent cell’ they came from (Figure 1A).
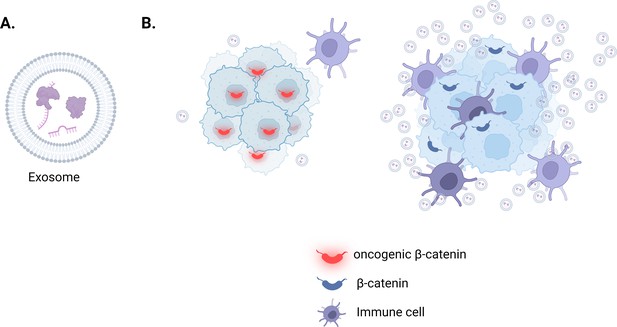
The role of exosomes in liver cancers associated with oncogenic β-catenin.
(A) Exosomes are small, membrane-encapsulated vesicles that cells release into their environment. The content of each vesicle is reflective of the cell it came from and may include nucleic acids, proteins, metabolites and lipids (purple). (B) Hepatocellular carcinoma cells (light blue with dark blue outline), which carry a mutated version of β-catenin (also known as oncogenic β-catenin; red), secrete fewer exosomes due to downregulating the expression of genes required to build and release these vesicles (left). The lack of exosomes in the tumor microenvironment reduces the number of immune cells (purple) infiltrating these tumors compared to other types of liver cancer which express a non-mutated version of β-catenin (dark blue; right).
© 2024, BioRender Inc. Figure 1 was created using BioRender, and is published under a CC BY-NC-ND license. Further reproductions must adhere to the terms of this license.
When exosomes were first discovered in 1981, they were thought to just be another mechanism cells use to relieve themselves of unwanted materials (Trams et al., 1981; Johnstone et al., 1987; Harding et al., 1983; Kucharzewska et al., 2013). However, more recently exosomes have emerged as important players in how cells communicate with each other, even when they are far apart. Exosomes released from the parent cell are taken up by or fused into the membrane of a recipient cell. This delivery of active biological material can influence the behavior of the recipient cell, such as the genes it expresses and how it responds to stimuli (Valadi et al., 2007; Chen et al., 2014).
In cancer, for example, exosomes released from tumor cells have been shown to promote drug resistance and the local formation of blood vessels (Hu et al., 2015; Chen et al., 2014; Qu et al., 2016). They can also mediate how cancer cells interact with the immune system, such as impairing how immune cells detect and target cancerous cells (Liu et al., 2020; Kalluri and LeBleu, 2020; Ning et al., 2018; Mathieu et al., 2019). Despite these advances, however, much remains unknown about the role exosomes play in cancer and, importantly, what regulates exosome secretion in the parent cell. Now, in eLife, Clotilde Billottet, Violaine Moreau and co-workers – including Camille Dantzer as first author – report how mutations in the gene for β-catenin affect how liver cancer cells secrete exosomes and, in turn, the recruitment of immune cells to the tumor (Dantzer et al., 2024).
Many tumors carry mutations in the gene for β-catenin, including around 30–40% of tumors associated with a type of liver cancer called hepatocellular carcinoma (de L Coste et al., 1998; Rebouissou et al., 2016). This group of tumors has an important unifying feature: they cannot be infiltrated by cells of the immune system, and are therefore resistant to immunotherapy. But it was unclear what was causing this effect.
To investigate, Dantzer et al. (who are based at the University of Bordeaux and University of Limoges) studied a cell-based model of hepatocellular carcinoma in which the mutant β-catenin (also known as oncogenic β-catenin) had been silenced. They found that this increased the expression of genes involved in generating and secreting exosomes (Figure 1B). It also restored the number of exosomes secreted from the cancer cells, suggesting that oncogenic β-catenin represses the secretion of these vesicles.
To better understand the molecular mechanism at play, Dantzer et al. focused their study on two genes that are downregulated when oncogenic β-catenin is present: one of these genes codes for the small GTPase Rab27a, and the other codes for the receptor protein syndecan-4. Further experiments showed that these two genes are repressed in multiple cell models of liver cancer carrying oncogenic β-catenin. This correlation was also observed in tissue samples from patients with liver cancer, where the presence of oncogenic β-catenin corresponded with fewer transcripts of both Rab27a and syndecan-4.
Dantzer et al. also conducted experiments in a cell model of liver cancer in which β-catenin is not mutated. They found that activating the β-catenin pathway decreased the expression of the genes for Rab27a and syndecan-4, thus resulting in reduced exosome secretion. This suggests that β-catenin may be a master regulator of exosome machinery.
Finally, Dantzer et al. investigated the functional consequence of secreting fewer exosomes. To do this, they studied three dimensional models of hepatocellular carcinoma in which the gene for Rab27a had been depleted. The concomitant loss of exosomes resulted in a marked decrease in immune cells infiltrating the tumor models.
While it is still unclear how exosomes derived from liver cancer cells affect the immune system, this elegant study provides important mechanistic information about why immune cells struggle to infiltrate these tumors. However, there are many questions that remain to be answered. For instance, it is unclear if the exosomes secreted from cancer cells carrying oncogenic β-catenin contain the same materials as those secreted from cancer cells containing the non-mutated version of the gene. It is possible that the exosomes released from the cancer cells are transporting tumor-associated antigens that are required to stimulate an immune response. Perhaps most importantly it remains to be seen how general this mechanism of exosome regulation is, and whether β-catenin regulates exosome secretion in other contexts as well. Answering these questions may provide new treatment avenues for diseases in which exosome secretion has been blocked, or is harming tissues and cells.
References
-
Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytesThe Journal of Cell Biology 97:329–339.https://doi.org/10.1083/jcb.97.2.329
-
Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes)The Journal of Biological Chemistry 262:9412–9420.
-
Exosomes: From garbage bins to translational medicineInternational Journal of Pharmaceutics 583:119333.https://doi.org/10.1016/j.ijpharm.2020.119333
-
Exfoliation of membrane ecto-enzymes in the form of micro-vesiclesBiochimica et Biophysica Acta 645:63–70.https://doi.org/10.1016/0005-2736(81)90512-5
Article and author information
Author details
Publication history
- Version of Record published: May 14, 2024 (version 1)
Copyright
© 2024, Schmieder
This article is distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use and redistribution provided that the original author and source are credited.
Metrics
-
- 468
- views
-
- 66
- downloads
-
- 0
- citations
Views, downloads and citations are aggregated across all versions of this paper published by eLife.
Download links
Downloads (link to download the article as PDF)
Open citations (links to open the citations from this article in various online reference manager services)
Cite this article (links to download the citations from this article in formats compatible with various reference manager tools)
Further reading
-
- Cancer Biology
- Immunology and Inflammation
Solid tumors generally exhibit chromosome copy number variation, which is typically caused by chromosomal instability (CIN) in mitosis. The resulting aneuploidy can drive evolution and associates with poor prognosis in various cancer types as well as poor response to T-cell checkpoint blockade in melanoma. Macrophages and the SIRPα-CD47 checkpoint are understudied in such contexts. Here, CIN is induced in poorly immunogenic B16F10 mouse melanoma cells using spindle assembly checkpoint MPS1 inhibitors that generate persistent micronuclei and diverse aneuploidy while skewing macrophages toward a tumoricidal ‘M1-like’ phenotype based on markers and short-term anti-tumor studies. Mice bearing CIN-afflicted tumors with wild-type CD47 levels succumb similar to controls, but long-term survival is maximized by SIRPα blockade on adoptively transferred myeloid cells plus anti-tumor monoclonal IgG. Such cells are the initiating effector cells, and survivors make de novo anti-cancer IgG that not only promote phagocytosis of CD47-null cells but also suppress tumor growth. CIN does not affect the IgG response, but pairing CIN with maximal macrophage anti-cancer activity increases durable cures that possess a vaccination-like response against recurrence.
-
- Cancer Biology
The tumor microenvironment is a determinant of cancer progression and therapeutic efficacy, with nutrient availability playing an important role. Although it is established that the local abundance of specific nutrients defines the metabolic parameters for tumor growth, the factors guiding nutrient availability in tumor compared to normal tissue and blood remain poorly understood. To define these factors in renal cell carcinoma (RCC), we performed quantitative metabolomic and comprehensive lipidomic analyses of tumor interstitial fluid (TIF), adjacent normal kidney interstitial fluid (KIF), and plasma samples collected from patients. TIF nutrient composition closely resembles KIF, suggesting that tissue-specific factors unrelated to the presence of cancer exert a stronger influence on nutrient levels than tumor-driven alterations. Notably, select metabolite changes consistent with known features of RCC metabolism are found in RCC TIF, while glucose levels in TIF are not depleted to levels that are lower than those found in KIF. These findings inform tissue nutrient dynamics in RCC, highlighting a dominant role of non-cancer-driven tissue factors in shaping nutrient availability in these tumors.






